what happens when solute is added to a supersaturated solution
13.2: Saturated Solutions and Solubility
- Page ID
- 21785
Learning Objectives
- To empathize the relationship between solubility and molecular construction.
- To demonstrate how the forcefulness of intramolecular bonding determines the solubility of a solute in a given solvent.
When a solute dissolves, its individual atoms, molecules, or ions collaborate with the solvent, become solvated, and are able to diffuse independently throughout the solution (Effigy \(\PageIndex{1a}\)). This is non, however, a unidirectional process. If the molecule or ion happens to collide with the surface of a particle of the undissolved solute, it may attach to the particle in a process called crystallization. Dissolution and crystallization continue as long every bit excess solid is nowadays, resulting in a dynamic equilibrium analogous to the equilibrium that maintains the vapor pressure of a liquid. We can represent these opposing processes every bit follows:
\[ solute + solvent \underset{crystallization}{\stackrel{dissolution}{\longrightleftharpoons}} solution \]
Although the terms precipitation and crystallization are both used to describe the separation of solid solute from a solution, crystallization refers to the formation of a solid with a well-defined crystalline structure, whereas atmospheric precipitation refers to the formation of whatsoever solid phase, oftentimes ane with very pocket-sized particles.

Factors Affecting Solubility
The maximum amount of a solute that tin can dissolve in a solvent at a specified temperature and pressure is its solubility. Solubility is often expressed as the mass of solute per book (g/L) or mass of solute per mass of solvent (g/chiliad), or as the moles of solute per volume (mol/L). Even for very soluble substances, however, there is usually a limit to how much solute tin can deliquesce in a given quantity of solvent. In general, the solubility of a substance depends on not simply the energetic factors we have discussed just also the temperature and, for gases, the pressure. At 20°C, for case, 177 g of NaI, 91.2 g of NaBr, 35.9 yard of NaCl, and merely 4.1 g of NaF deliquesce in 100 1000 of water. At 70°C, nonetheless, the solubilities increase to 295 g of NaI, 119 g of NaBr, 37.5 1000 of NaCl, and 4.8 one thousand of NaF. As you learned in Chapter 12, the lattice energies of the sodium halides increase from NaI to NaF. The fact that the solubilities decrease every bit the lattice free energy increases suggests that the \(ΔH_2\) term in Figure 13.ane dominates for this series of compounds.
A solution with the maximum possible amount of solute is saturated. If a solution contains less than the maximum amount of solute, information technology is unsaturated. When a solution is saturated and excess solute is present, the rate of dissolution is exactly equal to the rate of crystallization (Figure \(\PageIndex{1b}\)). Using the value only stated, a saturated aqueous solution of NaCl, for instance, contains 35.9 g of NaCl per 100 mL of water at 20°C. We can fix a homogeneous saturated solution by calculation backlog solute (in this case, greater than 35.nine g of NaCl) to the solvent (water), stirring until the maximum possible amount of solute has dissolved, and so removing undissolved solute by filtration.
The solubility of most solids increases with increasing temperature.
Because the solubility of most solids increases with increasing temperature, a saturated solution that was prepared at a college temperature unremarkably contains more dissolved solute than it would contain at a lower temperature. When the solution is cooled, it can therefore get supersaturated (Effigy \(\PageIndex{1c}\)). Like a supercooled or superheated liquid, a supersaturated solution is unstable. Consequently, adding a small particle of the solute, a seed crystal, volition normally cause the excess solute to rapidly precipitate or crystallize, sometimes with spectacular results. The charge per unit of crystallization in Equation \(\ref{13.2.1}\) is greater than the charge per unit of dissolution, so crystals or a precipitate form (Figure \(\PageIndex{1d}\)). In contrast, adding a seed crystal to a saturated solution reestablishes the dynamic equilibrium, and the net quantity of dissolved solute no longer changes.
Video \(\PageIndex{1}\): hot ice (sodium acetate) cute science experiment. watered-downwardly sodium acetate trihydrate. Needle crystal is truly wonderful structures
Considering crystallization is the opposite of dissolution, a substance that requires an input of heat to class a solution (\(ΔH_{soln} > 0\)) releases that oestrus when it crystallizes from solution (\(ΔH_{crys} < 0\)). The amount of heat released is proportional to the amount of solute that exceeds its solubility. Two substances that take a positive enthalpy of solution are sodium thiosulfate (\(Na_2S_2O_3\)) and sodium acetate (\(CH_3CO_2Na\)), both of which are used in commercial hot packs, small bags of supersaturated solutions used to warm hands (see Figure 13.i.3).
Interactions in Liquid Solutions
The interactions that determine the solubility of a substance in a liquid depend largely on the chemic nature of the solute (such equally whether it is ionic or molecular) rather than on its physical state (solid, liquid, or gas). We will start describe the general case of forming a solution of a molecular species in a liquid solvent and and so describe the formation of a solution of an ionic chemical compound.
Solutions of Molecular Substances in Liquids
The London dispersion forces, dipole–dipole interactions, and hydrogen bonds that concur molecules to other molecules are mostly weak. Fifty-fifty so, energy is required to disrupt these interactions. As described in Section 13.ane, unless some of that free energy is recovered in the formation of new, favorable solute–solvent interactions, the increase in entropy on solution formation is not enough for a solution to form.
For solutions of gases in liquids, we can safely ignore the energy required to divide the solute molecules (\(ΔH_2 = 0\)) because the molecules are already separated. Thus we need to consider only the energy required to separate the solvent molecules (\(ΔH_1\)) and the free energy released past new solute–solvent interactions (\(ΔH_3\)). Nonpolar gases such every bit \(N_2\), \(O_2\), and \(Ar\) have no dipole moment and cannot engage in dipole–dipole interactions or hydrogen bonding. Consequently, the only mode they can interact with a solvent is by means of London dispersion forces, which may be weaker than the solvent–solvent interactions in a polar solvent. It is non surprising, then, that nonpolar gases are virtually soluble in nonpolar solvents. In this case, \(ΔH_1\) and \(ΔH_3\) are both small and of similar magnitude. In dissimilarity, for a solution of a nonpolar gas in a polar solvent, \(ΔH_1\) is far greater than \(ΔH_3\). Every bit a result, nonpolar gases are less soluble in polar solvents than in nonpolar solvents. For case, the concentration of \(N_2\) in a saturated solution of \(N_2\) in water, a polar solvent, is only \(7.07 \times ten^{-4}\; One thousand\) compared with \(iv.5 \times ten^{-3}\; M\) for a saturated solution of \(N_2\) in benzene, a nonpolar solvent.
The solubilities of nonpolar gases in h2o generally increase as the molecular mass of the gas increases, as shown in Tabular array \(\PageIndex{ane}\). This is precisely the trend expected: as the gas molecules get larger, the forcefulness of the solvent–solute interactions due to London dispersion forces increases, budgeted the strength of the solvent–solvent interactions.
| Gas | Solubility (Thou) × 10−4 |
|---|---|
| He | 3.90 |
| Ne | 4.65 |
| Ar | 15.2 |
| Kr | 27.9 |
| Xe | 50.2 |
| H2 | 8.06 |
| Due northtwo | 7.07 |
| CO | 10.six |
| O2 | 13.9 |
| N2O | 281 |
| CHiv | fifteen.5 |
Virtually all mutual organic liquids, whether polar or not, are miscible. The strengths of the intermolecular attractions are comparable; thus the enthalpy of solution is expected to be small (\(ΔH_{soln} \approx 0\)), and the increment in entropy drives the germination of a solution. If the predominant intermolecular interactions in two liquids are very different from ane another, however, they may be immiscible. For instance, organic liquids such as benzene, hexane, \(CCl_4\), and \(CS_2\) (S=C=South) are nonpolar and have no ability to act as hydrogen bond donors or acceptors with hydrogen-bonding solvents such as \(H_2O\), \(HF\), and \(NH_3\); hence they are immiscible in these solvents. When shaken with water, they form carve up phases or layers separated by an interface (Figure \(\PageIndex{2}\)), the region between the two layers.

Just because 2 liquids are immiscible, however, does not mean that they are completely insoluble in each other. For example, 188 mg of benzene dissolves in 100 mL of h2o at 23.5°C. Adding more benzene results in the separation of an upper layer consisting of benzene with a pocket-size amount of dissolved water (the solubility of water in benzene is merely 178 mg/100 mL of benzene). The solubilities of simple alcohols in water are given in Tabular array \(\PageIndex{2}\).
| Alcohol | Solubility (mol/100 g of \(H_2O\)) |
|---|---|
| methanol | completely miscible |
| ethanol | completely miscible |
| n-propanol | completely miscible |
| n-butanol | 0.11 |
| due north-pentanol | 0.030 |
| n-hexanol | 0.0058 |
| n-heptanol | 0.0008 |
Only the three lightest alcohols (methanol, ethanol, and n-propanol) are completely miscible with water. As the molecular mass of the alcohol increases, and then does the proportion of hydrocarbon in the molecule. Correspondingly, the importance of hydrogen bonding and dipole–dipole interactions in the pure booze decreases, while the importance of London dispersion forces increases, which leads to progressively fewer favorable electrostatic interactions with water. Organic liquids such as acetone, ethanol, and tetrahydrofuran are sufficiently polar to be completely miscible with water nevertheless sufficiently nonpolar to be completely miscible with all organic solvents.
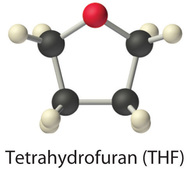
The aforementioned principles govern the solubilities of molecular solids in liquids. For instance, elemental sulfur is a solid consisting of cyclic \(S_8\) molecules that have no dipole moment. Because the \(S_8\) rings in solid sulfur are held to other rings by London dispersion forces, elemental sulfur is insoluble in h2o. It is, however, soluble in nonpolar solvents that take comparable London dispersion forces, such as \(CS_2\) (23 g/100 mL). In contrast, glucose contains five –OH groups that can form hydrogen bonds. Consequently, glucose is very soluble in water (91 g/120 mL of water) but essentially insoluble in nonpolar solvents such every bit benzene. The structure of one isomer of glucose is shown here.

Low-molecular-mass hydrocarbons with highly electronegative and polarizable element of group vii atoms, such as chloroform (\(CHCl_3\)) and methylene chloride (\(CH_2Cl_2\)), have both pregnant dipole moments and relatively stiff London dispersion forces. These hydrocarbons are therefore powerful solvents for a wide range of polar and nonpolar compounds. Naphthalene, which is nonpolar, and phenol (\(C_6H_5OH\)), which is polar, are very soluble in chloroform. In dissimilarity, the solubility of ionic compounds is largely adamant not by the polarity of the solvent only rather by its dielectric constant, a mensurate of its ability to separate ions in solution, as y'all volition shortly see.
Instance \(\PageIndex{1}\)
Identify the most important solute–solvent interactions in each solution.
- iodine in benzene solvent
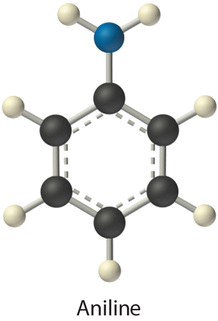
- aniline (\(\ce{C_6H_5NH_2}\)) in dichloromethane (\(\ce{CH_2Cl_2}\)) solvent
- iodine in water solvent
Given: components of solutions
Asked for: predominant solute–solvent interactions
Strategy:
Identify all possible intermolecular interactions for both the solute and the solvent: London dispersion forces, dipole–dipole interactions, or hydrogen bonding. Decide which is probable to be the most important factor in solution formation.
Solution
- Benzene and \(\ce{I2}\) are both nonpolar molecules. The only possible attractive forces are London dispersion forces.
- Aniline is a polar molecule with a dipole moment of one.six D and has an \(\ce{–NH_2}\) group that tin human action as a hydrogen bond donor. Dichloromethane is also polar with a 1.5 D dipole moment, but it has no obvious hydrogen bail acceptor. Therefore, the nigh important interactions between aniline and \(CH_2Cl_2\) are probable to be dipole-dipole interactions.
- Water is a highly polar molecule that engages in extensive hydrogen bonding, whereas \(I_2\) is a nonpolar molecule that cannot act every bit a hydrogen bond donor or acceptor. The slight solubility of \(\ce{I_2}\) in water (\(i.3 \times x^{-3}\; mol/L\) at 25°C) is due to London dispersion forces.
Exercise \(\PageIndex{1}\)
Place the most of import interactions in each solution:
- ethylene glycol (\(HOCH_2CH_2OH\)) in acetone
- acetonitrile (\(\ce{CH_3C≡N}\)) in acetone
- n-hexane in benzene
- Answer a
-
hydrogen bonding
- Reply b
-
London interactions
- Answer c
-
London dispersion forces
Hydrophilic and Hydrophobic Solutes
A solute can be classified as hydrophilic (literally, "water loving"), significant that information technology has an electrostatic attraction to water, or hydrophobic ("water fearing"), significant that information technology repels h2o. A hydrophilic substance is polar and often contains O–H or North–H groups that can form hydrogen bonds to water. For example, glucose with its five O–H groups is hydrophilic. In dissimilarity, a hydrophobic substance may exist polar simply commonly contains C–H bonds that do not interact favorably with water, as is the case with naphthalene and due north-octane. Hydrophilic substances tend to be very soluble in water and other strongly polar solvents, whereas hydrophobic substances are essentially insoluble in water and soluble in nonpolar solvents such as benzene and cyclohexane.
The difference between hydrophilic and hydrophobic substances has substantial consequences in biological systems. For example, vitamins can be classified as either fat soluble or water soluble. Fatty-soluble vitamins, such equally vitamin A, are more often than not nonpolar, hydrophobic molecules. As a result, they tend to be absorbed into fat tissues and stored there. In contrast, water-soluble vitamins, such as vitamin C, are polar, hydrophilic molecules that broadcast in the blood and intracellular fluids, which are primarily aqueous. Water-soluble vitamins are therefore excreted much more rapidly from the trunk and must exist replenished in our daily diet. A comparing of the chemical structures of vitamin A and vitamin C quickly reveals why one is hydrophobic and the other hydrophilic.
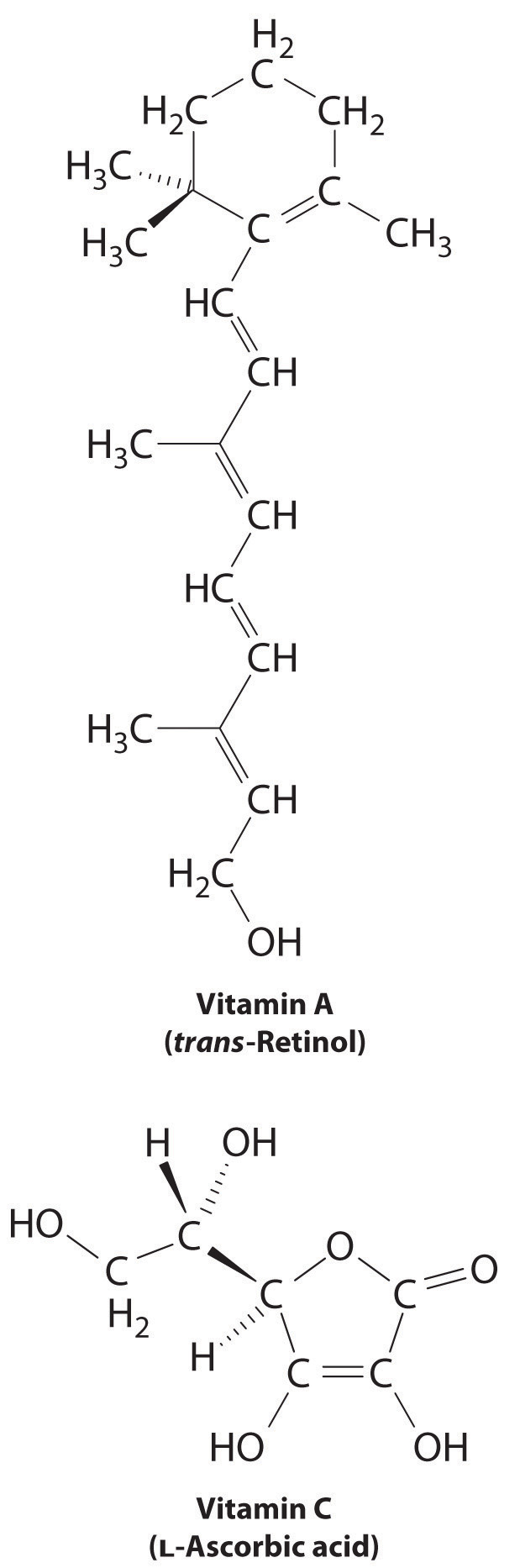
Considering water-soluble vitamins are rapidly excreted, the risk of consuming them in excess is relatively small. Eating a dozen oranges a day is likely to make you tired of oranges long before y'all suffer whatsoever ill effects due to their high vitamin C content. In contrast, fat-soluble vitamins establish a significant wellness take chances when consumed in large amounts. For example, the livers of polar bears and other large animals that alive in cold climates contain large amounts of vitamin A, which have occasionally proven fatal to humans who have eaten them.
Instance \(\PageIndex{2}\)
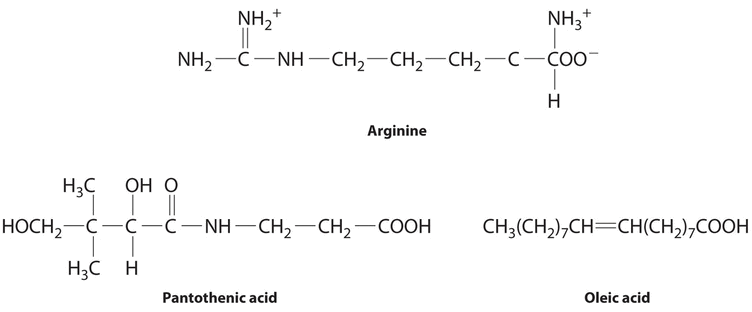
The following substances are essential components of the human diet:
Using what you know of hydrophilic and hydrophobic solutes, classify each as water soluble or fat soluble and predict which are likely to be required in the diet on a daily ground.
- arginine
- pantothenic acid
- oleic acid
Given: chemical structures
Asked for: classification as water soluble or fatty soluble; dietary requirement
Strategy:
Based on the structure of each compound, decide whether it is hydrophilic or hydrophobic. If it is hydrophilic, it is likely to exist required on a daily basis.
Solution:
- Arginine is a highly polar molecule with two positively charged groups and one negatively charged group, all of which tin can form hydrogen bonds with water. As a issue, it is hydrophilic and required in our daily nutrition.
- Although pantothenic acrid contains a hydrophobic hydrocarbon portion, it too contains several polar functional groups (\(\ce{–OH}\) and \(\ce{–CO_2H}\)) that should interact strongly with h2o. Information technology is therefore probable to be h2o soluble and required in the nutrition. (In fact, pantothenic acrid is almost always a component of multiple-vitamin tablets.)
- Oleic acid is a hydrophobic molecule with a single polar group at one cease. It should be fat soluble and not required daily.
Exercise \(\PageIndex{2}\)
These compounds are consumed by humans: caffeine, acetaminophen, and vitamin D. Identify each as primarily hydrophilic (water soluble) or hydrophobic (fat soluble), and predict whether each is likely to be excreted from the body speedily or slowly.

- Answer
-
Caffeine and acetaminophen are water soluble and rapidly excreted, whereas vitamin D is fatty soluble and slowly excreted
Solid Solutions
Solutions are not limited to gases and liquids; solid solutions also be. For instance, amalgams, which are normally solids, are solutions of metals in liquid mercury. Because about metals are soluble in mercury, amalgams are used in aureate mining, dentistry, and many other applications. A major difficulty when mining gold is separating very pocket-sized particles of pure gilded from tons of crushed rock. One fashion to attain this is to agitate a suspension of the crushed rock with liquid mercury, which dissolves the gold (as well as any metal silverish that might be present). The very dumbo liquid gold–mercury amalgam is then isolated and the mercury distilled away.
An blend is a solid or liquid solution that consists of i or more elements in a metal matrix. A solid blend has a single homogeneous phase in which the crystal structure of the solvent remains unchanged by the presence of the solute. Thus the microstructure of the alloy is uniform throughout the sample. Examples are substitutional and interstitial alloys such equally brass or solder. Liquid alloys include sodium/potassium and gold/mercury. In contrast, a partial alloy solution has two or more than phases that can be homogeneous in the distribution of the components, but the microstructures of the two phases are not the aforementioned. As a liquid solution of lead and tin is cooled, for example, different crystalline phases form at different cooling temperatures. Alloys usually take properties that differ from those of the component elements.
Network solids such as diamond, graphite, and \(\ce{SiO_2}\) are insoluble in all solvents with which they do not react chemically. The covalent bonds that hold the network or lattice together are simply too strong to be broken under normal conditions. They are certainly much stronger than any believable combination of intermolecular interactions that might occur in solution. Most metals are insoluble in well-nigh all solvents for the same reason: the delocalized metallic bonding is much stronger than any favorable metallic cantlet–solvent interactions. Many metals react with solutions such equally aqueous acids or bases to produce a solution. However, every bit we saw in Section 13.1, in these instances the metal undergoes a chemic transformation that cannot be reversed by simply removing the solvent.
Solids with very stiff intermolecular bonding tend to be insoluble.
Solubilities of Ionic Substances in Liquids
Previously, you were introduced to guidelines for predicting the solubility of ionic compounds in water. Ionic substances are generally most soluble in polar solvents; the higher the lattice free energy, the more polar the solvent must be to overcome the lattice energy and deliquesce the substance. Because of its high polarity, h2o is the most mutual solvent for ionic compounds. Many ionic compounds are soluble in other polar solvents, however, such as liquid ammonia, liquid hydrogen fluoride, and methanol. Because all these solvents consist of molecules that have relatively big dipole moments, they tin can collaborate favorably with the dissolved ions.
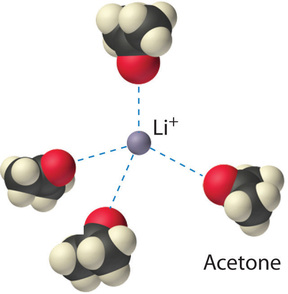
The ion–dipole interactions between \(\ce{Li^{+}}\) ions and acetone molecules in a solution of LiCl in acetone are shown in Figure \(\PageIndex{three}\). The energetically favorable \(\ce{Li^{+}}\)–acetone interactions make \(ΔH_3\) sufficiently negative to overcome the positive \(ΔH_1\) and \(ΔH_2\). Because the dipole moment of acetone (ii.88 D), and thus its polarity, is actually larger than that of water (one.85 D), one might fifty-fifty look that LiCl would be more soluble in acetone than in water. In fact, the opposite is truthful: 83 k of LiCl dissolve in 100 mL of h2o at xx°C, but only about iv.1 g of \(\ce{LiCl}\) dissolve in 100 mL of acetone. This credible contradiction arises from the fact that the dipole moment is a holding of a single molecule in the gas phase. A more useful mensurate of the ability of a solvent to dissolve ionic compounds is its dielectric constant (ε), which is the ability of a bulk substance to decrease the electrostatic forces between ii charged particles. By definition, the dielectric constant of a vacuum is 1. In essence, a solvent with a high dielectric abiding causes the charged particles to behave as if they have been moved further apart. At 25°C, the dielectric abiding of water is fourscore.one, i of the highest known, and that of acetone is simply 21.0. Hence h2o is meliorate able to decrease the electrostatic allure betwixt \(\ce{Li^{+}}\) and \(\ce{Cl^{-}}\) ions, and so \(\ce{LiCl}\) is more than soluble in water than in acetone. This behavior is in contrast to that of molecular substances, for which polarity is the dominant gene governing solubility.
A solvent's dielectric constant is the most useful measure of its power to dissolve ionic compounds. A solvent's polarity is the dominant factor in dissolving molecular substances.
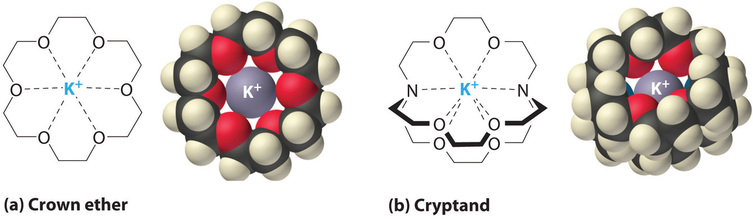
It is also possible to deliquesce ionic compounds in organic solvents using crown ethers, cyclic compounds with the general formula \((OCH_2CH_2)_n\). Crown ethers are named using both the total number of atoms in the ring and the number of oxygen atoms. Thus 18-crown-6 is an 18-membered band with vi oxygen atoms (Figure \(\PageIndex{1a}\)). The cavity in the center of the crown ether molecule is lined with oxygen atoms and is large plenty to be occupied by a cation, such every bit \(K^+\). The cation is stabilized by interacting with lone pairs of electrons on the surrounding oxygen atoms. Thus crown ethers solvate cations inside a hydrophilic cavity, whereas the outer crush, consisting of C–H bonds, is hydrophobic. Crown ethers are useful for dissolving ionic substances such as \(KMnO_4\) in organic solvents such as isopropanol \([(CH_3)_2CHOH]\) (Figure \(\PageIndex{five}\)). The availability of crown ethers with cavities of different sizes allows specific cations to be solvated with a loftier caste of selectivity.

Cryptands (from the Greek kryptós, meaning "hidden") are compounds that can completely surround a cation with lonely pairs of electrons on oxygen and nitrogen atoms (Figure \(\PageIndex{4b}\)). The number in the name of the cryptand is the number of oxygen atoms in each strand of the molecule. Similar crown ethers, cryptands tin can be used to ready solutions of ionic compounds in solvents that are otherwise besides nonpolar to dissolve them.
Summary
The solubility of a substance is the maximum amount of a solute that can dissolve in a given quantity of solvent; it depends on the chemic nature of both the solute and the solvent and on the temperature and pressure. When a solution contains the maximum amount of solute that can dissolve nether a given fix of conditions, it is a saturated solution. Otherwise, it is unsaturated. Supersaturated solutions, which contain more dissolved solute than allowed under detail conditions, are not stable; the addition of a seed crystal, a small particle of solute, will ordinarily cause the excess solute to crystallize. A system in which crystallization and dissolution occur at the same charge per unit is in dynamic equilibrium. The solubility of a substance in a liquid is determined by intermolecular interactions, which also make up one's mind whether two liquids are miscible. Solutes can be classified every bit hydrophilic (water loving) or hydrophobic (water fearing). Vitamins with hydrophilic structures are water soluble, whereas those with hydrophobic structures are fat soluble. Many metals dissolve in liquid mercury to form amalgams. Covalent network solids and most metals are insoluble in near all solvents. The solubility of ionic compounds is largely determined by the dielectric constant (ε) of the solvent, a measure of its ability to subtract the electrostatic forces betwixt charged particles. Solutions of many ionic compounds in organic solvents tin be dissolved using crown ethers, cyclic polyethers large enough to accommodate a metallic ion in the center, or cryptands, compounds that completely surround a cation.
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/13%3A_Properties_of_Solutions/13.2%3A_Saturated_Solutions_and_Solubility
0 Response to "what happens when solute is added to a supersaturated solution"
Post a Comment